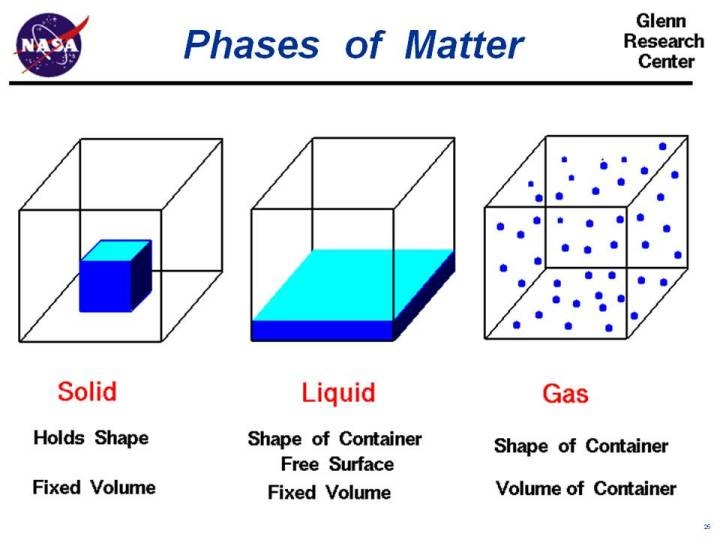
What's the matter?
~ Exploring Chemical Science ~
Firing Up the Brain:
Brainstorm: List five objects under the following three categories:
a) Solid:
b) Liquid:
c) Gases:
Brainstorm: List five objects under the following three categories:
a) Solid:
b) Liquid:
c) Gases:
Preparing the Brain:
Learning Goal: Be able to independently describe what a solid, liquid and gas is.
Social Goal: Work as a class, in group, in paris and individually displaying the iValue "I think clearly"
Engaging the Brain:
Watch the following informative video:
Learning Goal: Be able to independently describe what a solid, liquid and gas is.
Social Goal: Work as a class, in group, in paris and individually displaying the iValue "I think clearly"
Engaging the Brain:
Watch the following informative video:
Firing Up the Brain:
Interactive Instructions: Working in groups to classify objects four objects on the list as a solid, liquid or gas.
1. stones:
2. icing sugar:
3. blue-tack
4. elastic bands
Interactive Instructions: Working in groups to classify objects four objects on the list as a solid, liquid or gas.
1. stones:
2. icing sugar:
3. blue-tack
4. elastic bands
Thinking Strategies: Complete the last four object classification independently and then confirm and check with your group.
5. cooking oil
6. honey
7. air
8. hairspray (tricky!)
5. cooking oil
6. honey
7. air
8. hairspray (tricky!)
Independent Consolidation and Practice: Complete one of the following interactive games:
Checking and Recording Understanding: How would you describe solids, liquids and gases? Write and/or draw your thoughts:
Solids are...
Liquids are...
Gases are...
Strengthening the Brain:
Confirming your classifications of the eight items.
Personal Reflection:
a) Which object was the hardest to classify?
b) Which classification was the hardest to explain?
Solids are...
Liquids are...
Gases are...
Strengthening the Brain:
Confirming your classifications of the eight items.
Personal Reflection:
a) Which object was the hardest to classify?
b) Which classification was the hardest to explain?
See how they run
- recognising that substances exist in different states depending on the temperature
- observing that gases have mass and take up space, demonstrated by using balloons or bubble
- exploring the way solids, liquids and gases change under different situations such as heating and cooling
- recognising that not all substances can be easily classified on the basis of their observable properties
Firing Up the Brain:
1. Brainstrom as many liquids as possible.
2. Circle the liquids you think DON'T have water in them in red.
3. Put a green line under the liquids that are thick.
1. Brainstrom as many liquids as possible.
2. Circle the liquids you think DON'T have water in them in red.
3. Put a green line under the liquids that are thick.
Prepare the Brain:
Learning Goals:
* Explore and record in a table the properties of different liquids
* Identify properties that are shared by liquids
* Discuss how liquids flow and take the shape of their containers
Social Goal Focus:
* I am responsible
Learning Goals:
* Explore and record in a table the properties of different liquids
* Identify properties that are shared by liquids
* Discuss how liquids flow and take the shape of their containers
Social Goal Focus:
* I am responsible
Engaging the Brain:
Which of these are liquids? Give three reasons to outline how you can scientifically explain if it is a liquid or not?
e.g Our claim is that _____________ is/is not a liquid. Our evidence is __________________________.
Which of these are liquids? Give three reasons to outline how you can scientifically explain if it is a liquid or not?
e.g Our claim is that _____________ is/is not a liquid. Our evidence is __________________________.
How to test the objects:
~ turn the container upside down
~ shake the container
~ use a magnifying glass to look carefully
~tipping the container and seeing how long substances take to flow to the other end
Hint: Liquids can be described as sticky, smooth, wet and gooey.
~ turn the container upside down
~ shake the container
~ use a magnifying glass to look carefully
~tipping the container and seeing how long substances take to flow to the other end
Hint: Liquids can be described as sticky, smooth, wet and gooey.
Stretching the Brain:
Science Starter Questions:
Asking for Evidence
~ I have a questions about ________________________ .
~ How does your evidence support you claim ____________________?
~ What other evidence do you have to support your claim ______________________?
Agreeing/Disagree
I agree with ____________________ because __________________.
I disagree with _______________________ because _____________________.
One difference between my idea and yours is _______________________.
Questioning further
I wonder what would happen if ______________________?
What caused _______________________?
I wonder why __________________________?
How would it be different if _______________________?
What do you think would happen if _____________________?
Science Starter Questions:
Asking for Evidence
~ I have a questions about ________________________ .
~ How does your evidence support you claim ____________________?
~ What other evidence do you have to support your claim ______________________?
Agreeing/Disagree
I agree with ____________________ because __________________.
I disagree with _______________________ because _____________________.
One difference between my idea and yours is _______________________.
Questioning further
I wonder what would happen if ______________________?
What caused _______________________?
I wonder why __________________________?
How would it be different if _______________________?
What do you think would happen if _____________________?
Strengthening the Brain:
1. The common properties of liquids that we found are: ______________________________.
2. Some of the things that are different between liquids are: _________________________.
3. We had difficulty describing _______________________ because _______________________.
1. The common properties of liquids that we found are: ______________________________.
2. Some of the things that are different between liquids are: _________________________.
3. We had difficulty describing _______________________ because _______________________.
What a Gas!
- recognising that substances exist in different states depending on the temperature
- observing that gases have mass and take up space, demonstrated by using balloons or bubble
- exploring the way solids, liquids and gases change under different situations such as heating and cooling
- recognising that not all substances can be easily classified on the basis of their observable properties
Fire Up the Brain:
Brainstorm:
1. Can you give some examples of any common gases.
2. Underline the gases that you can smell in blue
http://www.mikecurtis.org.uk/ks2_gases.htm
Brainstorm:
1. Can you give some examples of any common gases.
2. Underline the gases that you can smell in blue
http://www.mikecurtis.org.uk/ks2_gases.htm
Prepare the Brain:
Learning Goal:
~ identify that air is made up of gases and that it takes up space.
~ work in a collaborative learning team to change one variable in a fair test investigation about air.
~ compare air and water and discuss how gases spread out to fill their container
Social Goal:
~ I make good choices
Learning Goal:
~ identify that air is made up of gases and that it takes up space.
~ work in a collaborative learning team to change one variable in a fair test investigation about air.
~ compare air and water and discuss how gases spread out to fill their container
Social Goal:
~ I make good choices
Engaging the Brain:
~ Is a balloon a solid that surrounds a gas. Here are some other examples of a gas inside a solid, can you think of any others?
~ Is a balloon a solid that surrounds a gas. Here are some other examples of a gas inside a solid, can you think of any others?
Stretching the Brain:
An empty cup... Is it really empty?
You will need a copy of the "Tissues in a Cup" experiment.
An empty cup... Is it really empty?
You will need a copy of the "Tissues in a Cup" experiment.
Strengthening the Brain
What conclusions can you draw from this experiment?
If I had a balloon filled with air, where would the air go if I untied it?
If I had a balloon filled with water, where would the water go if I untied it?
What conclusions can you draw from this experiment?
If I had a balloon filled with air, where would the air go if I untied it?
If I had a balloon filled with water, where would the water go if I untied it?
Independant Work to make you a super genius
Listed below is a number of activities to help you in your Science learning. Some of these will act as scientific revision, some will teach you something you didn't previously know. Your responsibility is to list the game in your science book and write down three facts that were covered in the game.
Videos
Hot Stuff
- recognising that substances exist in different states depending on the temperature
- observing that gases have mass and take up space, demonstrated by using balloons or bubble
- exploring the way solids, liquids and gases change under different situations such as heating and cooling
- recognising that not all substances can be easily classified on the basis of their observable properties
What is a solid?
What is a liquid?
What is a gas?
What is a liquid?
What is a gas?
Can you define the following scientific terms:
Solid
Compressed
Melting
Evaporation
Collision
Pressure
Condensation
Freezing
Sublimation
Looking at the screenshots below, what state is each photo showing and what is the process to get there? The first one is done for you:
Solid
Compressed
Melting
Evaporation
Collision
Pressure
Condensation
Freezing
Sublimation
Looking at the screenshots below, what state is each photo showing and what is the process to get there? The first one is done for you:
Choose one of the pictures below and outline the following:
1. What change is the picture showing?
2. What is this change called?
3. Why has this change happened?
4. What are happening to the atoms/particles for this to happen?
1. What change is the picture showing?
2. What is this change called?
3. Why has this change happened?
4. What are happening to the atoms/particles for this to happen?